KYOTO.- Nearly all the chemical energy available to Earth's lifeforms can be traced back to the sun. This is because light-harvesting (LH) supramolecules (two or more molecules held together by intermolecular forces) enable plants and some types of bacteria (typically at the base of the food chain) to leverage sunlight for driving photosynthesis. For these supramolecules to be effective, they need to have multiple pigments, such as chlorophyll, arranged in special structures that vary among species.
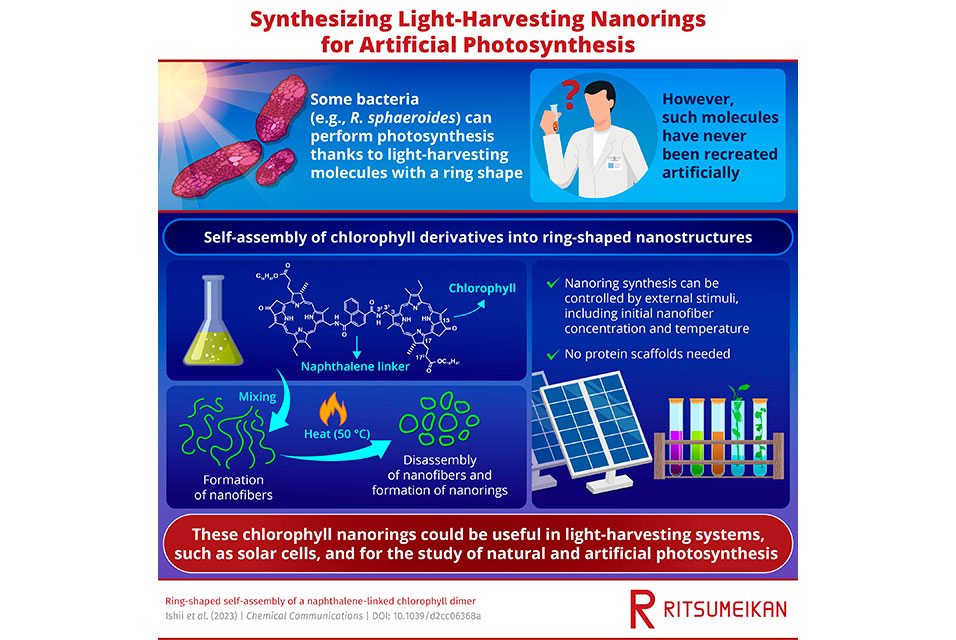
For instance, green photosynthetic bacteria have LH antennas in which chlorophyll molecules form spiral structures that, in turn, aggregate into large tubular supramolecules. In contrast, purple photosynthetic bacteria, such as Rhodobacter sphaeroides, exhibit different types of LH antennas in which chlorophyll pigments are arranged into ring-shaped architectures. While researchers have managed to recreate the tubular chlorophyl aggregates in the lab with a self-assembly approach, their ring-shaped counterparts have been not artificially reproduced so far.
In a recent study published in Chemical Communications, a team of scientists from Japan managed to address this knowledge gap. They discovered that mixing a chlorophyll derivative with naphthalenediamide in an organic solvent led to the formation of dimers that spontaneously self-assembled into ring-shaped structures, each several hundred nanometers in diameter. The team included Professor Hitoshi Tamiaki from
Ritsumeikan University and Assistant Professor Shogo Matsubara from Nagoya Institute of Technology.
Surprised by their initial discovery, the team sought to better understand the formation of the ring-shaped nanostructures and their properties. Upon closer inspection using atomic force microscopy, they observed that chlorophyll dimers, molecules composed of two chlorophyll units linked by naphthalene, initially self-assembled into stable wavy nanofibers. Upon heating these nanofibers at 50°C, they disassembled into smaller nanoring precursors whose ends eventually joined together to form the desired nanorings.
Interestingly, this nanofiber–nanoring transformation was dependent on external stimuli. Temperature was observed to play a major role, as well as dimer concentration. Prof. Tamiaki explains, "At low concentrations, ring-shaped aggregates were obtained by a preferential end-to-end joining of a single fiber supramolecule. In contrast, end-to-end linkage between different nanofibers was prevalent at higher concentrations and gave rise to network nanostructures."
Overall, the findings of this study reveal a straightforward way to synthesize the LH supramolecule that has eluded scientists for a long time. Excited about the results, Dr. Matsubara remarks, "The self-assemblies we synthesized enable efficient sunlight absorption along with excitation energy migration and transfer. Mimicking the arrangement of chlorophyll pigments observed in nature is critical to not only understand natural photosynthesis but also construct artificial LH systems for devices such as solar cells."
Moreover, the structural change from nanofiber to nanoring triggered by external stimuli could help realize novel smart materials with tunable properties. The team said that further investigations on the optical properties of the self-assembled nanorings are underway.