ZURICH.- Cancer patients might one day benefit from being administered immune cells from healthy donors. But as things stand, receiving donor cells can cause severe or even fatal immune reactions. A researcher at
ETH Zurich has now developed a technology that avoids these.
Edo Kapetanovic is a medical doctor, but for a while now he has devoted himself entirely to research in synthetic immunology. He has completed his doctoral studies in immunoengineering and is working at the Department of Biosystems Science and Engineering at ETH Zurich in Basel. His big goal is to develop new cancer therapies by providing patients with immune cells derived from donor blood. He is now getting closer to this goal: he has managed to modify donor cells so that they attack only the tumour cells and not patient’s healthy cells. The technology has been tested in the lab in human cells, but it will take more time and development before the patients can benefit from the technology.
Administering donor cells is far from straightforward: the immune system is specialised in distinguishing foreign molecules from ‘self’ and will attack any foreign cell. This is particularly dangerous for immunocompromised patients, as donor cells can recognize patient cells as foreign and trigger a violent and, consequently, fatal immune response in the recipient, known as a graft-versus-host reaction. That is why today’s immunotherapeutic treatments for cancer mainly use a patient’s own immune cells rather than donated cells.
Kapetanovic and his team have now succeeded in engineering immune cells that are safe of graft-versus-host reaction.
A body’s own cells don’t always work
Generally speaking, approved immunotherapies for cancer take one of two approaches, and both depend on cells known as killer cells, mostly killer T cells. In one approach, experts extract the patient’s own killer cells and modify them in the lab so that they specifically recognise and eliminate cancer cells. These modified cells are then administered to the patient.
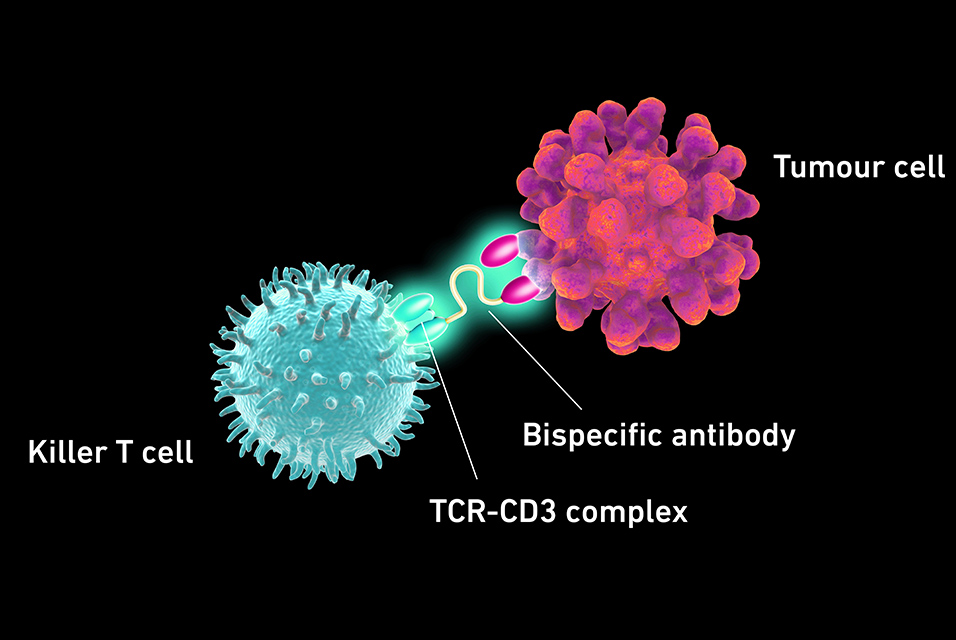
The second approach uses what are known as bispecific antibodies as medications. In the body, these provide a molecular link between killer T cells and cancer cells, thereby activating the former to fight the tumour cells.
However, these approaches have a serious disadvantage, as Kapetanovic explains: “Both approaches make use of the patient’s own cells. But it’s a known fact that immune cells differ greatly from one person to the next. In some patients, the T cells simply aren’t fit enough to fight the tumour.” So it would be helpful if cancer patients could be supplied with powerful killer T cells from healthy donors – but the dangerous graft-versus-host reaction stands in the way. Kapetanovic has now solved this problem for the approach that uses bispecific antibodies.
To understand the challenge the researcher faced, it helps to know the following: the desired activation of killer T cells by bispecific antibodies and the undesired activation by the bystander healthy cells, leading to the graft-versus-host reaction, occurs via the same molecular complex on the outer membrane of the killer T cells, the TCR-CD3 complex.
Cells with artificial protein complex
Kapetanovic was able to decouple the desired from the undesired activation by creating a synthetic TCR-CD3 complex. Killer T cells with this synthetic complex are no longer able to respond to foreign cells, which means they can no longer trigger a graft-versus-host reaction. Nevertheless, these killer T cells can still be activated with bispecific antibodies to fight cancer cells.
The synthetic TCR-CD3 complex is the result of elaborate and detailed studies by Kapetanovic and his team. Before the scientists could specifically modify this complex, they extensively studied the molecular structure of each TCR-CD3 complex subunits and how they transmit the activation signal.
Combination therapy with cells and antibodies
“Our approach aims to one day offer a standardised, off-the-shelf product for cancer therapy,” Kapetanovic says. The idea is to isolate the killer T cells from blood of healthy donors and use biotechnical methods to replace their natural TCR-CD3 complex with the synthetic one. Cancer patients would then have their blood enriched with these modified donor cells before going on to be treated with bispecific antibodies.
“This would make it possible to administer cells from any donor to any patient,” Kapetanovic says. Such a standardised product would be much easier and cheaper to produce than having to isolate and modify cells from each individual patient, as is the case today. The current process is laborious and requires complex laboratory infrastructure, which limits the availability of treatment to patients. An off-the-shelf product could be more easily manufactured and distributed, meaning more patients could benefit.
ETH Zurich has filed a patent application for the new technology. Kapetanovic now plans to develop it further and bring it to market. To this end, he is receiving financial support from the Swiss Innovation Agency Innosuisse and plans to found a spin-off company.