SÃO PAULO.- In an article published in the journal Nature Communications, researchers at the
University of São Paulo in Brazil show how protozoans of the genus Leishmania that cause leishmaniasis manipulate a protein that plays an essential role in the organism's defense in order to continue to replicate, preventing the body from vanquishing the disease.
The findings of the study reported in the article offer hope for the development of novel treatments for the disease. Some 30,000 new cases of leishmaniasis are notified annually, according to the World Health Organization (WHO), many of them in Brazil, and there are no specific medications or vaccines to combat the disease.
The protein in question is gasdermin-D, produced by macrophages and other cells in the human innate immune system (the first to join battle when a pathogen is detected). It induces an inflammatory process required to defend the organism against infectious agents such as bacteria and parasites.
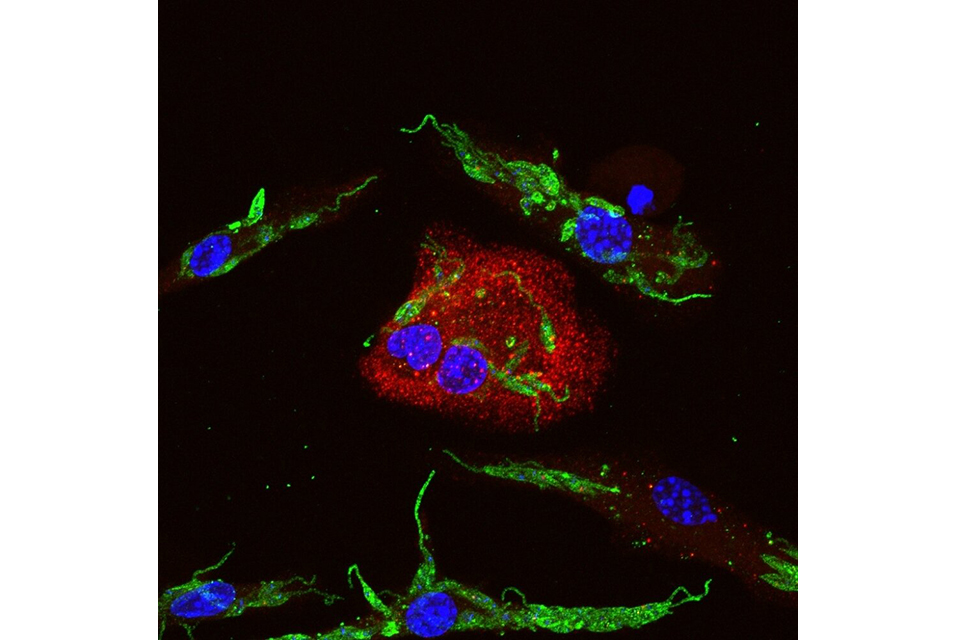
"Gasdermin-D is important to activation of the inflammasome, a complex of proteins involved in the organism's defense against infection. We observed activation of the inflammasome in biopsies from patients with tegumental leishmaniasis [comprising the cutaneous and mucocutaneous forms]," said Keyla de Sá, first author of the article. The study was conducted during her Ph.D. research at the Ribeirão Preto Medical School (FMRP-USP). She is currently a postdoctoral researcher at Yale University in the United States.
In experiments with macrophages and mice infected by Leishmania, gasdermin-D activation was too weak to promote the cell death required to combat the parasite, and the inflammation therefore persisted, she explained.
The inflammatory process is responsible for the appearance of the lesions caused by the disease, leading to scars and deformations, and even physical incapacitation, depending on the part of the body affected.
The experiments showed that the parasite manages to bring about alternative cleavage of gasdermin-D, changing the protein's structural form and inactivating it so that it cannot perform its inflammatory functions. In other infections, gasdermin-D is cleaved by macrophage proteins, causing cell death and preventing the infectious agents from continuing to replicate.
"It's very interesting to see how this parasite modulates the functions of macrophages, which are cells that specialize in killing microbes. The process enables Leishmania to remain in mammalian hosts for years, sometimes for the entire lifetime of an infected individual," said Dario Zamboni, principal investigator for the study and last author of the article.
Inflammasome
Inflammasomes trigger inflammation to combat infectious agents. The article by the researchers at FMRP-USP analyzes the role played by the protein NLRP3 in mediating the inflammasome. The NLRP3 inflammasome is critical for host immune defenses against bacterial, fungal and viral infections but has been linked to the pathogenesis of several inflammatory disorders when dysregulated.
Previous research by the group investigated the role of the inflammasome in severe COVID-19 cases, when it is overactivated and creates a cytokine storm, which can lead to death. They also successfully tested a drug that inhibits NLRP3 inflammasome activation in animals and human cells and could in future be given to severe COVID-19 patients.
"We now have the data to test the same drug or another drug on patients with severe tegumental leishmaniasis, who have highly exacerbated inflammation. However, caution is necessary because in less severe cases the inflammatory process triggered by the inflammasome can be important to control the disease," Zamboni explained.
The tests involved four of the most common species that cause tegumental leishmaniasis: Leishmania amazonensis, L. mexicana, L. major and L. braziliensis.
Tegumental and visceral leishmaniasis, which attacks several internal organs, is one of 20 neglected tropical diseases that mainly affect the poor in Africa, Asia and Latin America. In 2020, the WHO announced a plan to eliminate or eradicate some of these and drastically reduce the incidence of others by 2030. Among other initiatives, the plan calls for the development of novel drugs, since the few existing medications have toxic side effects and patients tend not to take them for long enough.